‘Our own front line’: Ukrainian surgeons see wave of wounded soldiers since counteroffensive began
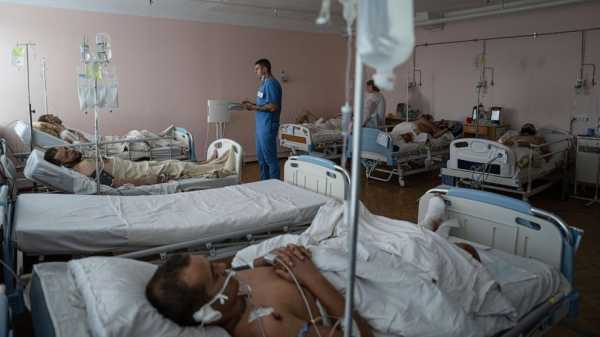
DNIPRO, Ukraine — The horrors of war arrive through the night at a hospital in eastern Ukraine, a procession of stretchers bearing limp bodies whisked from the front line. The soldiers come with bandaged limbs soaked in blood, faces blackened…








